Covid-19 prevention drug now available for at-risk patients
The electronic platform taking applications for a drug that helps prevent Covid-19 in people who are at a particular risk from the disease opened on Thursday.
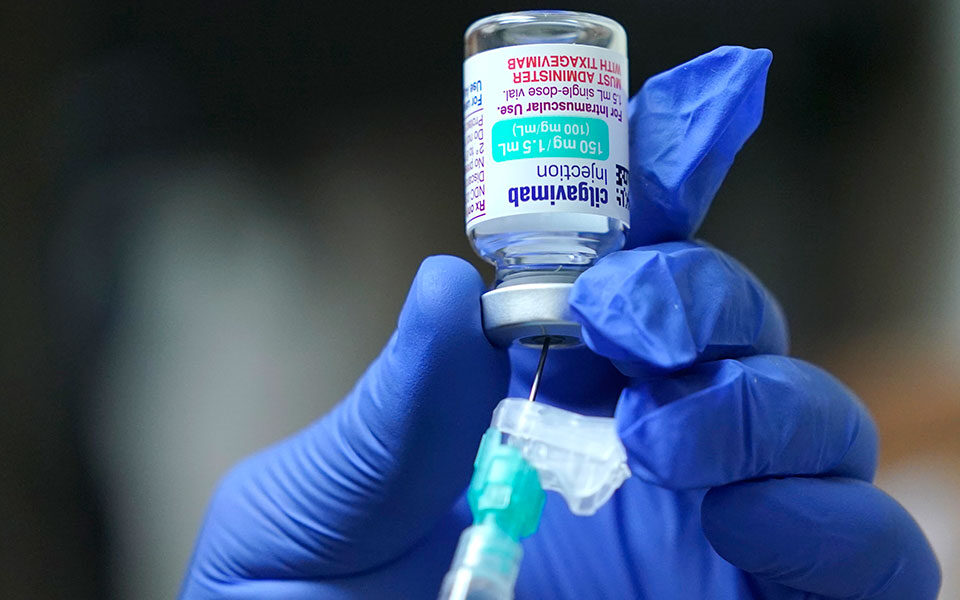
According to the Health Ministry, the monoclonal antibody medicine developed by AstraZeneca and called Evusheld, has been approved by the European Medicines Agency and is now available in Greece.
The medicine is approved for adults and adolescents from 12 years of age, weighing at least 40 kilograms, who have compromised immune systems and are also at risk from the Covid vaccine.
They also need to be completely clear of the SARS-CoV-2 virus, without having been infected for at least three months.
The application for the two-injection dose of Evusheld needs to be submitted by the patient’s doctor, along with evidence supporting why he or she needs the medicine instead of a vaccine.
Doses can be administered at hospitals only, the ministry’s circular said.