Greek drug watchdog dismisses reports on suspension of Astrazeneca vaccine batch
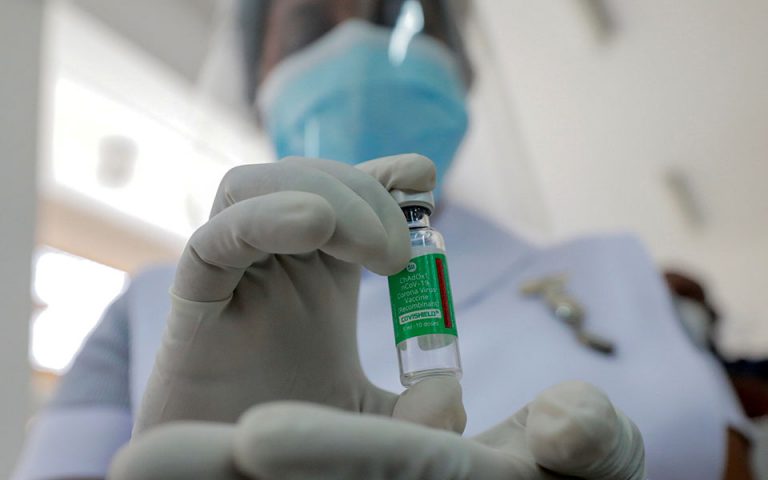
Greece’s pharmaceutical regulator (EOF) rejected local press reports on Wednesday that it had suspended a batch of the Oxford/AstraZeneca coronavirus vaccine after Austrian authorities announced they were investigating a person’s death and another’s illness after taking the jab.
Austrian authorities clarified on March 7 that there was currently “no evidence” that the reactions were linked to the vaccination, but that remaining stocks of the batch will no longer be used as a precaution.
“EOF informs that they [the reports] do not correspond to reality. The European Medicines Agency, the manufacturer, as well as the Austrian Medicines Agency, do not associate this batch and vaccination with the adverse reactions recorded in Austria,” the National Organization of Medicines said in a press release on Wednesday.
The Latvian Health Inspectorate and the State Agency of Medicines (ZVA) also suspended batch ABV5300 of AstraZeneca’s Covid-19 vaccine, following Austria’s example.
Latvian news agency LETA reported on March 9 that Batch ABV5300, which totals 1,6 million doses, has been delivered to 17 EU member states: Lithuania, Estonia, Luxembourg, Latvia, Ireland, Denmark, Bulgaria, Austria, Greece, Sweden, the Netherlands, Poland, Spain, France, Iceland, Malta and Cyprus.